Double Donation IVF: Step-by-Step Process for International Patients
Double donation IVF — a cycle using both donor eggs and donor sperm — offers one of the highest success rates available in assisted reproduction. For international patients, the process may feel complex at first glance, but with the right clinic it follows a clear, well-organized path. Below is a detailed guide to what you can expect: medically, logistically, and legally.

What Is Double Donation IVF?
Double donation IVF combines an egg donor and a sperm donor to create embryos that are transferred to the recipient's uterus. Neither partner contributes genetic material, yet the recipient carries the pregnancy and gives birth. This approach is recommended when both ovarian reserve and sperm quality are severely compromised, or when both partners carry genetic conditions that should not be passed on. Additionally, this type of treatment is suggested to single women who cannot use their own eggs.
Current data show clinical pregnancy rates of 55–65% per embryo transfer in optimized double-donation programs, depending on embryo quality and endometrial preparation protocol. [1][2]
Who Is This Option For?
Double donation is typically considered in the following situations:

- Both partners have poor gamete quality — severe male factor infertility combined with diminished ovarian reserve or premature ovarian insufficiency
- Recurrent IVF failures with own eggs and sperm, after thorough investigation of euploid embryo transfer journals
- High genetic risk on both sides — hereditary conditions in both partners. Requires a validation from a clinical geneticist as other approaches (such as PGT-M) may actually be the first choice strategy
- Single recipients who require both egg and sperm donation
- Advanced maternal age (typically 41+) with AMH and FSH levels outside normal range
- Post-oncological patients after chemotherapy or radiation affecting gonadal function[3]
Step-by-Step Process
Step 1: Initial Consultation and Medical Assessment
The process begins with a remote consultation — via video call — with your reproductive endocrinologist. You will share medical records: previous IVF cycles, hormonal panels (AMH, FSH, LH, estradiol), semen analysis, and general health history. This step usually takes 1–3 days and allows the clinic to confirm candidacy for double donation without requiring immediate travel.
A full workup for the recipient includes: uterine cavity assessment, mammogram, infectious disease testing (HIV, hepatitis B/C, syphilis) etc. [4]


Step 2: Donor Selection
Your European clinic's donor database will include anonymous, thoroughly screened donors — both egg and sperm. European clinics follow the guidelines established by the European Society of Human Reproduction and Embryology (ESHRE) [5], which require:
- Age limits: egg donors typically 18–35 years
- Genetic screening: karyotype, carrier testing for cystic fibrosis, SMA, fragile X, and expanded panels
- Infectious disease clearance and psychological evaluation
- Phenotype matching to recipient (appearance, blood group) upon request. However, access to the donor’s photos is never granted
You will receive a donor profile including physical characteristics, blood type, education background, and health history. Identity remains anonymous in most European jurisdictions, in compliance with local law.
Donor selection in Russia has specifics that may be considered a big advantage. In particular, there is no ban on access to donors’ photos. Clinics can show child and adult pictures of their donors without violating the law. Additionally, patients can negotiate a non-anonymous donor cycle where egg and sperm donors will become contactable upon the child's 18th birthday.
Step 3: Stimulation
Once donors and the recipient are confirmed, the clinical team launches egg donor stimulation. It will be followed by PGT-A of embryos and recipient endometrial preparation in the future.
The egg donor undergoes controlled ovarian stimulation (COS) for approximately 10–14 days, monitored by ultrasound and hormone levels. Trigger injection is administered when follicles reach appropriate size. Egg retrieval (oocyte pick-up) is performed under sedation. [6]
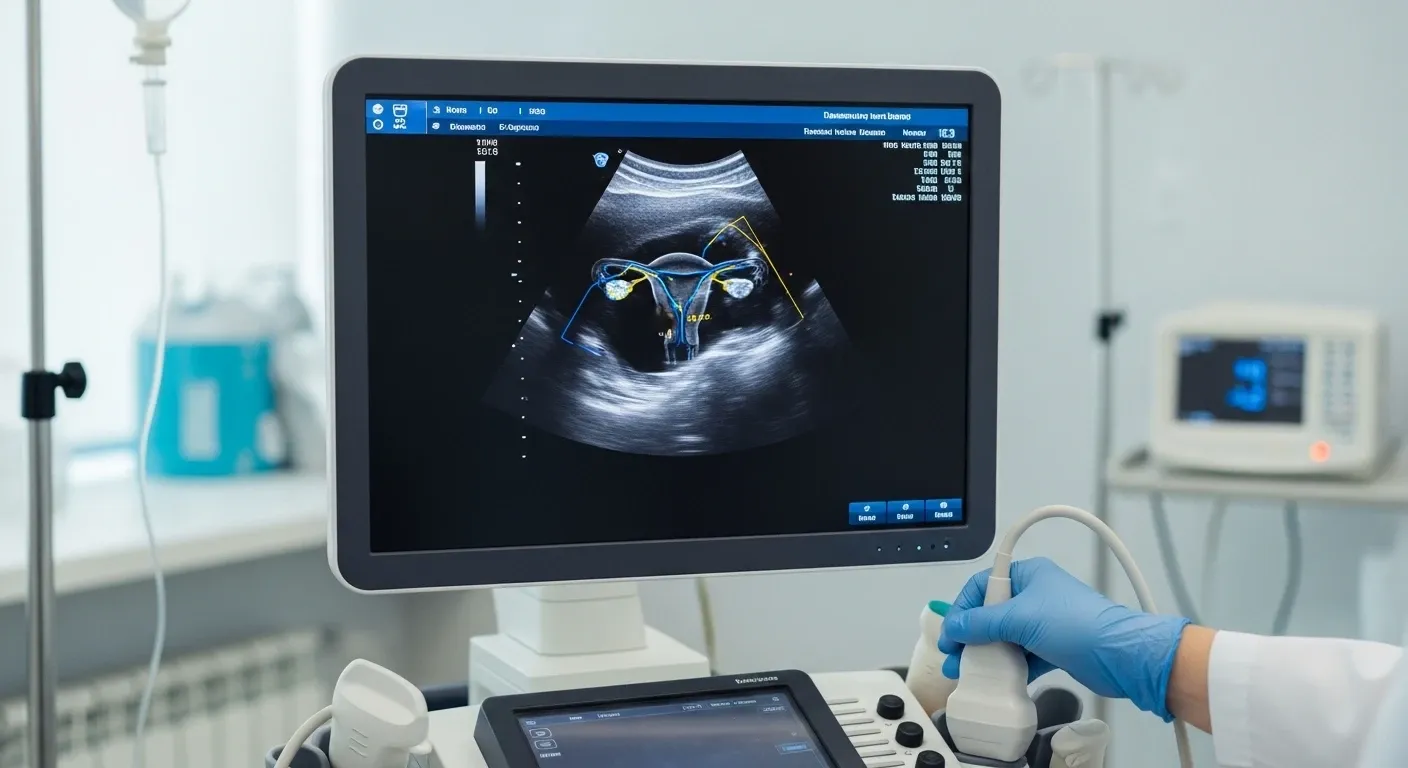
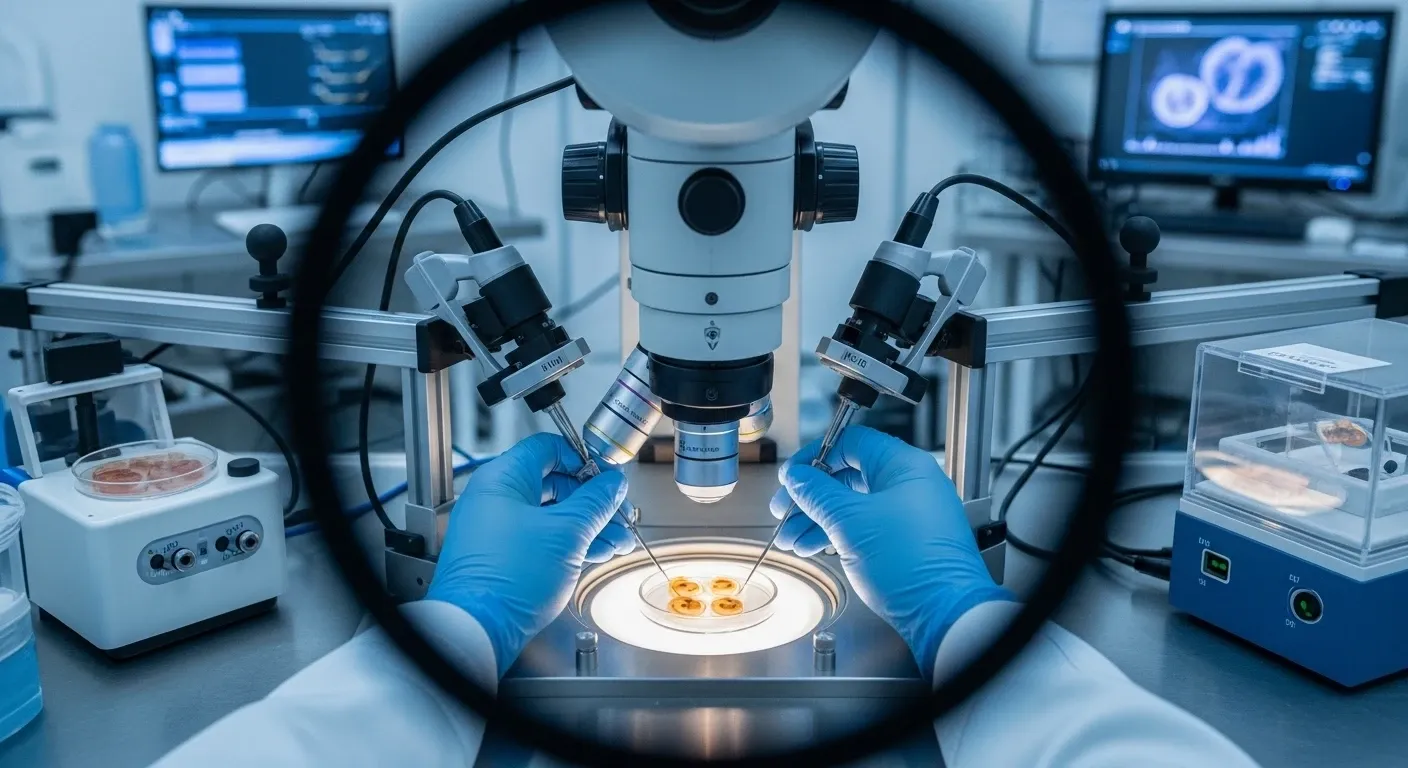
Step 4: Fertilization and Embryo Development
Retrieved oocytes are fertilized using the selected donor sperm via ICSI (intracytoplasmic sperm injection) — the standard technique in modern IVF that maximizes fertilization rates. Embryos develop in the laboratory for 5–7 days to the blastocyst stage, which is associated with significantly higher implantation potential compared to Day 3 transfers. [7]
Preimplantation Genetic Testing for Aneuploidy (PGT-A) is available upon request, allowing selection of chromosomally normal blastocysts and further improving success rates. [8]
Step 5: Endometrial Preparation and Your Travel
While embryos develop, the recipient prepares the uterine lining. A standard hormone replacement therapy (HRT) protocol begins with estradiol for 14-15 days, followed by progesterone supplementation when the endometrium reaches ≥7 mm with trilaminar pattern. This phase can be managed locally, by your own gynecologist, based on monitoring instructions from the treating clinic — minimizing travel time.
For the embryo transfer itself, most international patients need to be in the clinic for 4-5 days only. The transfer is a brief outpatient procedure requiring no anesthesia.

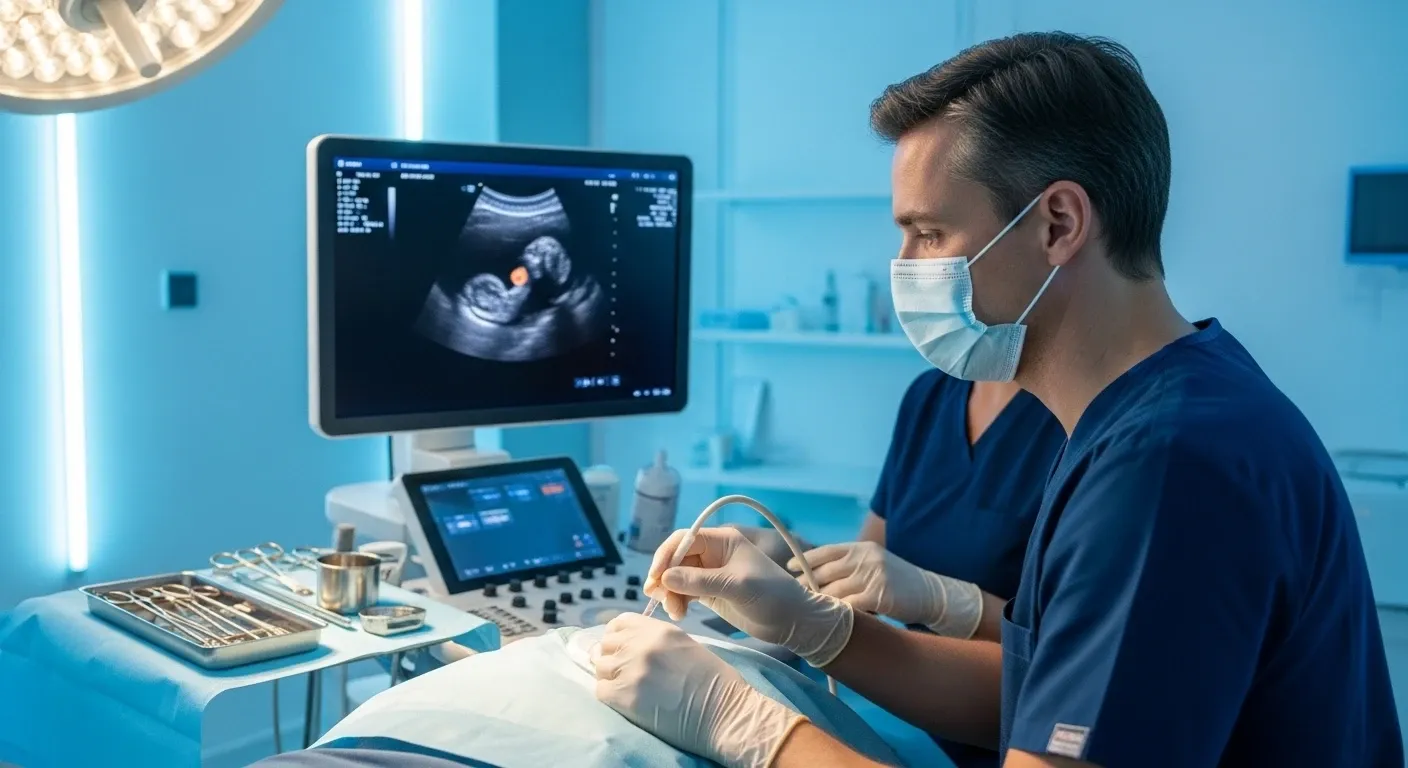
Step 6: Embryo Transfer
Transfer is performed under ultrasound guidance. A thin catheter carries a blastocyst into the uterine cavity. Most clinics favor single embryo transfer (SET) in line with ESHRE recommendations to minimize multiple pregnancy risk. [5] Remaining quality embryos are vitrified (frozen) for future use.
Step 7: Post-Transfer Support and Pregnancy Test
Progesterone and estrogen supplementation continues for 10–12 weeks if pregnancy is confirmed. The first beta-hCG test is performed 9-10 days post-transfer. A positive result is followed by an early ultrasound at 5–6 weeks to confirm fetal heartbeat. After the first trimester, care is transferred to your local obstetrician.
Ongoing coordination with your international coordinator ensures continuity between clinic and local care team throughout the pregnancy.

IVF with EGG DONATION
As a woman ages, various fertility issues arise. By the age of 41-42, time is of the essence. Going for donor egg IVF with a reputable clinic is the best way forward.
Access adult photos to choose a verified donor from NGC's online database. Plan a program that requires only one visit for several days.
Request online-consultation
Typical Timeline
|
Phase |
Duration |
|
Initial consultation & workup |
1–3 weeks (remotely) |
|
Donor matching |
1–3 weeks |
|
Stimulation & fertilization with thawed donor sperm |
2–3 weeks (no travel required) |
|
Endometrial preparation |
2.5-3 weeks (locally or remotely) |
|
Travel & embryo transfer |
4-5 days in clinic |
|
Pregnancy test |
10 post-transfer |
Legal and Ethical Framework
Regulations for gamete donation vary by country. Many international patients choose to travel to destinations that comply with the European legal frameworks — such as Spain, Czech Republic, or Greece — where donation is anonymous [9], while other patients who require more freedom of choice go to the Russian Federation where rigorous healthcare standards [10] and patient-oriented framework align [11].
Key legal points for international patients:
- The birth mother (recipient) is recognized as the legal mother in most jurisdictions, including Russia
- Donor’s full anonymity is legally protected in countries like Spain and Czech Republic
- Significantly more information about your donors can be accessed in Russia
- Contracts with non-anonymous donors (if applicable) require signing additional legal documentation
What to Ask Your Clinic Before Starting
|
Essential Questions for Your First Consultation What is your clinic's clinical pregnancy rate per transfer in double donation cycles? How large is your clinic’s active donor database and what screening do donors undergo? What is the cost difference with PGT-A? Can endometrial preparation be monitored remotely? What technological advances can the clinic offer? What support is available in my language during the process? How are frozen embryos stored, and for how long? |
Summary
Double donation IVF is a well-established path to parenthood with high success rates and a clearly structured process. For international patients, the key advantage is that most of the cycle can be managed remotely, with clinic visits limited to 4-5 days. Choosing a clinic with highly-skilled clinicians, trained international coordinators, a transparent donor program, and rigorous embryological laboratory standards is essential to having a smooth experience.
Make an appointment
[1] Pellicer A, et al. "Cumulative live birth rates in oocyte donation cycles: the role of donor and recipient factors." Fertility and Sterility. 2020;114(4):787–796.
[2] Cobo A, et al. "Oocyte vitrification as an efficient option for elective fertility preservation." Fertility and Sterility. 2016;105(3):755–764.
[3] Domingo J, et al. "Ovarian response to controlled stimulation in cancer patients prior to fertility preservation." Reproductive BioMedicine Online. 2012;24(4):380–386.
[4] Griesinger G, et al. "Screening the uterine cavity prior to IVF: clinical value of hysteroscopy." Reproductive BioMedicine Online. 2018;36(6):614–621.
[5] ESHRE Guideline Group on Good Practice in IVF Labs. "Revised guidelines for good practice in IVF laboratories." Human Reproduction. 2016;31(4):685–686.
[6] Humaidan P, et al. "Oocyte maturation induction: current clinical practice." Human Reproduction Update. 2011;17(5):590–604.
[7] Papanikolaou EG, et al. "Blastocyst vs cleavage-stage embryo transfer: a meta-analysis of randomised controlled trials." Human Reproduction. 2008;23(1):91–99.
[8] Dahdouh EM, et al. "The impact of blastocyst biopsy and comprehensive chromosome screening technology on preimplantation genetic screening: a systematic review." Human Reproduction Update. 2015;21(4):451–473.
[9] European Commission. "Directive 2004/23/EC on standards of quality and safety for the donation, procurement, testing, processing, preservation, storage and distribution of human tissues and cells." Official Journal of the European Union. 2004.
[10] Ministry of Health of The Russian Federation “Order of July 31, 2020 no. 803n on the procedure for the use of assisted reproductive technologies, contraindications, and restrictions to their use””
https://normativ.kontur.ru/document?moduleId=1&documentId=373901#h2736
[11] IVF and Egg Donation in Russia. https://www.eggdonationfriends.com/ivf-egg-donation-country-russia/#elementor-toc__heading-anchor-1
