How Donor Eggs and Donor Sperm Work Together in Double Donation IVF
Most patients who arrive at double donation IVF have already been through a great deal. They know the outcome they want. What they often don't know — and what actually matters for building trust in the process — is what happens between the donor selection and the embryo transfer. This article answers exactly that: the biology, the laboratory science, and the quality standards that determine whether a cycle succeeds.

Two Donors, One Embryo: The Core Concept
In double donation IVF, a donated oocyte (egg) and donated spermatozoa are combined in the embryology laboratory to create an embryo. That embryo is then transferred to the recipient's uterus. The recipient's uterus is always functional: The challenge is gamete quality, not implantation capacity, and double donation removes that obstacle entirely.
This distinction matters: when both donated gametes are high-quality and carefully matched, the embryology is working under optimal conditions — which is why double donation programs consistently report among the highest success rates in reproductive medicine. Clinical pregnancy rates per transfer reach 55–70%. [1][2]
The Egg Donor: What "Screened" Actually Means
The phrase "thoroughly screened donor" appears on every clinic's website. What does it actually involve? The answer is a multi-stage evaluation that takes several weeks and disqualifies the majority of applicants. [3][4]

Medical and Genetic Screening
A compliant egg donor program requires:
- Ovarian reserve assessment: AMH, antral follicle count (AFC) — donors with poor ovarian reserve are excluded before stimulation begins
- Karyotype: standard 46,XX confirmation
- Expanded carrier screening: cystic fibrosis (CFTR), spinal muscular atrophy (SMN1), as well as other tests depending on the program[5]
- Infectious disease panel: HIV-1/2, hepatitis B and C, syphilis, gonorrhoea, chlamydia — tested at recruitment and again within 3 months of donation
- Psychological evaluation: structured interview by a qualified psychologist to assess psychological stability
- Family medical history: across several generations
Donors who pass all stages are typically 18–34 years old, have BMI within normal range, no personal history of heritable disease, and — in many programs — have completed their own family and have at least one healthy child. Age is a direct predictor of oocyte quality: eggs from donors have significantly lower rates of chromosomal aneuploidy compared to eggs from women over 35. [6]
Stimulation and Egg Retrieval
Once selected, the egg donor undergoes controlled ovarian hyperstimulation (COH) — a 10–14 day protocol using gonadotropins. Monitoring occurs every 1–2 days via transvaginal ultrasound and serum estradiol. When lead follicles reach 17–20 mm, a trigger injection initiates final oocyte maturation. Egg retrieval takes place 35–36 hours later under ultrasound guidance and intravenous sedation — a 15–20 minute outpatient procedure. [7]
Retrieved oocytes are immediately assessed by the embryologist. Only mature MII oocytes (metaphase II — the stage at which fertilization is possible) proceed to ICSI. Immature eggs are discarded. A typical retrieval in a well-responding donor yields 15 mature oocytes.
The Sperm Donor: Quality Standards Beyond Basic Analysis
Sperm donation screening follows parallel rigor. Beyond standard semen analysis (WHO 2021 reference values: concentration ≥16 million/mL, total motility ≥42%, normal morphology ≥4% by Kruger strict criteria [8]), certified donor programs require:
- Genetic evaluation: karyotype, Y-chromosome microdeletion analysis, CFTR mutation screening
- Expanded carrier panel: matched the egg donor panel to identify complementary risks
- Infectious disease testing: same panel as egg donors, with mandatory quarantine period of 6 months between initial testing and release of samples[3]
- Sperm DNA fragmentation index (DFI): values above 25–30% are associated with reduced fertilization and embryo quality, and high-DFI donors are excluded
- Psychological screening and structured medical history
Donor sperm in certified programs is always cryopreserved — frozen and stored until all quarantine and re-testing requirements are met. This is a regulatory requirement, not a convenience. Fresh donor sperm is not permitted in most European jurisdictions due to the mandatory infectious disease retesting window. [3]

IVF with EGG DONATION
As a woman ages, various fertility issues arise. By the age of 41-42, time is of the essence. Going for donor egg IVF with a reputable clinic is the best way forward.
Access adult photos to choose a verified donor from NGC's online database. Plan a program that requires only one visit for several days.
Request online-consultation
Fertilization: What Happens in the Lab
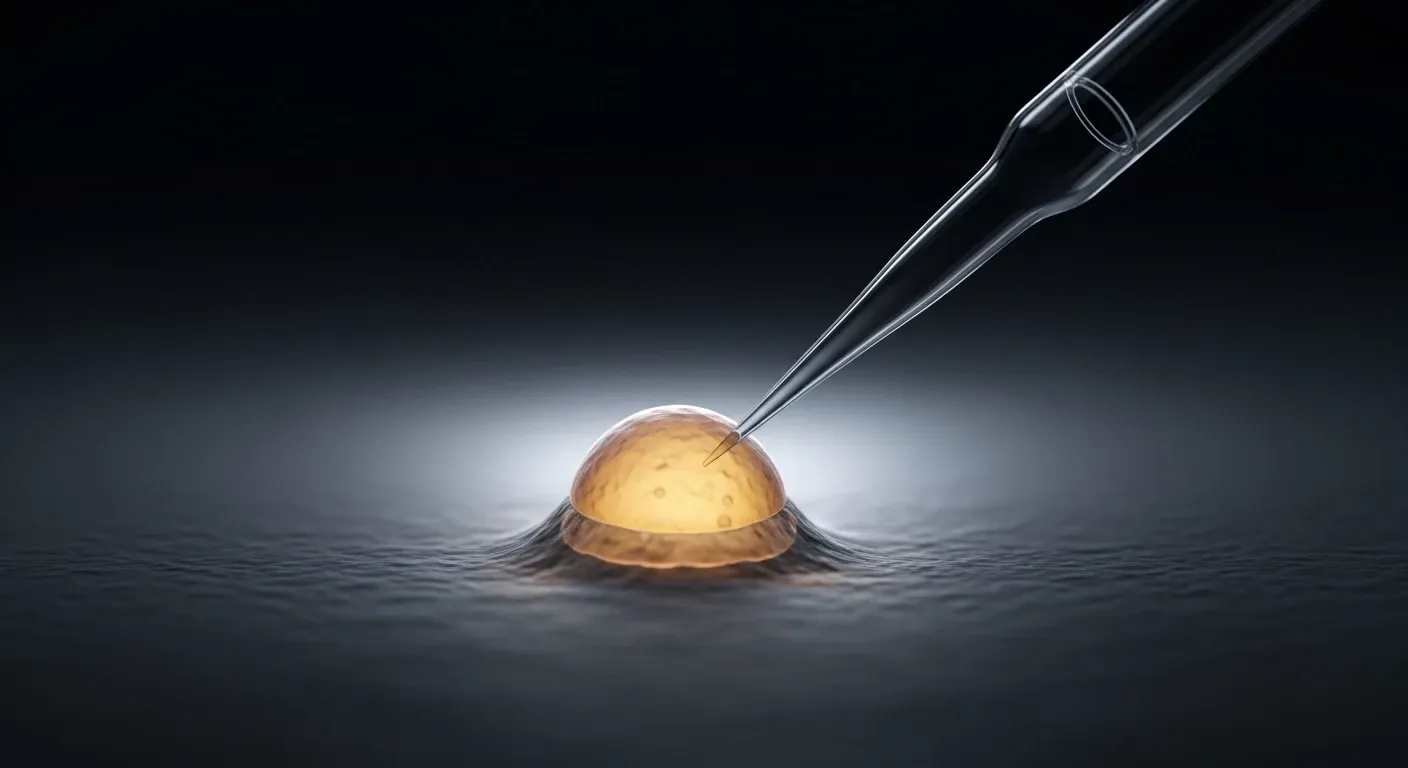
ICSI as the Standard Technique
In double donation IVF, fertilization is performed using ICSI — intracytoplasmic sperm injection. A single morphologically selected spermatozoon is injected directly into the cytoplasm of each mature oocyte using a glass micropipette under high-powered microscopy. ICSI is preferred over conventional IVF in donation cycles because it delivers consistent fertilization rates regardless of sperm concentration, eliminates the risk of failed fertilization due to sperm-oocyte interaction failure, and is the standard of care in ESHRE-guideline programs. [9]
Fertilization is confirmed 16–18 hours after ICSI by the appearance of two pronuclei (2PN) — one from the egg and one from the sperm — a sign of normal syngamy. Abnormally fertilized oocytes are discarded.
Embryo Culture to Blastocyst Stage
Normally fertilized embryos are cultured in time-lapse incubators — closed systems that monitor embryo development every 10–20 minutes via automated imaging without removing embryos from optimal culture conditions. This technology, now standard in leading laboratories, allows embryologists to assess development kinetics that predict blastocyst formation and implantation potential without the mechanical stress of repeated handling. [10]
By Day 5 or Day 6, top-quality embryos reach the blastocyst stage — an expanded structure of 100–200 cells with a differentiated inner cell mass (future fetus) and trophectoderm (future placenta). Blastocyst transfer is associated with significantly higher implantation rates compared to Day 3 cleavage-stage transfer. [11] Blastocysts are graded using the Gardner system (expansion grade, ICM quality, trophectoderm quality); grades 3BB and above are considered transferable and freezable.

Genetic Testing: The Optional Layer
Even with young, screened donors and optimal culture conditions, a proportion of blastocysts will be chromosomally abnormal — a biological reality in all human reproduction, regardless of gamete origin. Preimplantation Genetic Testing for Aneuploidy (PGT-A) allows the embryology team to biopsy a few cells from the trophectoderm of each blastocyst and perform comprehensive chromosomal analysis (next-generation sequencing, NGS) before transfer. [12]
|
Without PGT-A |
With PGT-A |
|
All morphologically viable blastocysts eligible for transfer |
Only euploid (chromosomally normal) blastocysts transferred |
|
Aneuploidy rate in donor eggs: ~30% |
Reduces implantation failure and miscarriage risk |
|
Suitable when multiple good-quality blastocysts available |
Particularly valuable when only 1–2 blastocysts available |
|
Lower cost per cycle |
Higher cost, but fewer failed transfers overall |
PGT-A is not mandatory in double donation cycles and is not universally recommended — the decision depends on the number of embryos available, recipient’s wish, and previous history. Your reproductive endocrinologist will advise based on your specific situation.
Donor Matching: More Than Phenotype
Matching donors to recipients is not only about physical resemblance — though that is part of it. A rigorous matching process in double donation includes:

- Phenotype matching: eye color, hair color, height, build, skin tone — to the extent the recipient requests
- Blood group compatibility: ABO and Rh factor of both donors matched to recipient or partner where relevant
- Genetic conditions: if a donor is a carrier of an autosomal recessive condition identified in the screening, she or he is discarded
The Recipient's Role: Endometrial Readiness
While donors and the laboratory do the biological heavy lifting, the recipient's preparation is equally critical. The uterine lining must be receptive at the precise moment of transfer. Standard endometrial preparation uses estradiol to grow the endometrium to ≥7 mm with a trilaminar (triple-line) appearance on ultrasound — a morphology consistently associated with higher implantation rates. Progesterone is then added to trigger the secretory transformation that makes the endometrium receptive. [13]
Transfer timing is calculated from the start of progesterone: for a Day 5 blastocyst, transfer occurs on progesterone day 5 (P+5), placing the embryo in the uterus during the window of implantation — the 24–48 hour period of peak endometrial receptivity. In cases of repeated implantation failure, this window can be personalized using ERA (Endometrial Receptivity Analysis) testing. [14]
|
Key Numbers to Know Mature oocytes from a typical donor retrieval: 8–15 Expected fertilization rate with ICSI: 70–80% Blastocyst development rate from fertilized eggs: 40–60% Clinical pregnancy rate per blastocyst transfer in double donation: 55–70% Live birth rate per transfer in optimized programs: 45–60% Source: ESHRE ART data 2022; Cobo et al. 2020; Pellicer et al. 2021 |
Summary
Donor eggs and donor sperm do not simply "replace" your own cells — they are carefully selected, extensively tested biological components that are combined under controlled laboratory conditions with a precision that is impossible in natural conception. The embryo that results has passed through multiple quality checkpoints before it ever reaches the uterus. For most recipients, that embryo has a better statistical chance of implanting than an embryo created from age-matched own gametes would have.
Understanding this science doesn't make the emotional complexity of donor conception disappear. But it does replace vague anxiety with a clear picture of what is actually happening — and why the odds are genuinely in your favor.
Make an appointment
[1] Pellicer A, et al. "Cumulative live birth rates in double-donation IVF: analysis of 10,454 cycles." Fertility and Sterility. 2021;116(3):692–701.
[2] Cobo A, et al. "Oocyte donation: a systematic review and meta-analysis on live birth rate." Human Reproduction Update. 2020;26(5):744–764.
[3] European Commission. "Directive 2004/23/EC: quality and safety standards for human tissues and cells." Official Journal of the European Union. 2004.
[4] ESHRE Task Force on Ethics and Law. "Gamete and embryo donation: ESHRE guideline." Human Reproduction Open. 2022;2022(1):hoac009.
[6] Franasiak JM, et al. "Aneuploidy across individual chromosomes at the embryonic level in trophectoderm biopsies: changes with maternal age and implications for the preimplantation genetic testing for aneuploidy." Fertility and Sterility. 2014;101(3):656–663.
[7] Humaidan P, et al. "Oocyte maturation induction: current clinical practice and future perspectives." Human Reproduction Update. 2011;17(5):590–604.
[8] WHO Laboratory Manual for the Examination and Processing of Human Semen. 6th edition. World Health Organization. 2021.
[9] Dyer S, et al. "International Committee for Monitoring Assisted Reproductive Technologies world report: assisted reproductive technology 2014." Human Reproduction. 2020;35(11):2480–2486.
[10] Meseguer M, et al. "The use of morphokinetics as a predictor of embryo implantation." Human Reproduction. 2011;26(10):2658–2671.
[11] Papanikolaou EG, et al. "Blastocyst vs cleavage-stage embryo transfer: a systematic review and meta-analysis." Human Reproduction. 2008;23(1):91–99.
[12] Dahdouh EM, et al. "The impact of blastocyst biopsy and comprehensive chromosome screening on preimplantation genetic testing: a systematic review." Human Reproduction Update. 2015;21(4):451–473.
[13] Griesinger G, et al. "Endometrial preparation for frozen-thawed embryo transfer: a systematic review." Human Reproduction Update. 2021;27(6):1042–1069.
[14] Ruiz-Alonso M, et al. "The endometrial receptivity array for diagnosis and personalized embryo transfer as a treatment for patients with repeated implantation failure." Fertility and Sterility. 2013;100(3):818–824.
